Part 11, ER/ES Guidelines
FDA 21 CFR Part 11
Part 11 is a regulation regarding electronic records and electronic signatures implemented by the U.S. Food and Drug Administration (FDA), which established requirements for transferring paper records to electronic media in 1997.
The provisions of Part 11 consist of a 32-page preamble and approximately three pages of the main text, which is configuration into the following three parts:
(1) General Provisions
(2) Electronic records
(3) Electronic signature
By applying Part 11, electronic records and electronic signatures will be treated as equivalent to paper records and handwritten signatures, but this is a legal expression and specific requirements require interpretation. The main requirements for devices and systems can be summarized as follows:
- Computerized System Validation
- Safety features
- Access restrictions (access management)
- Authenticity, legibility, duplication and protection of electronic records
- Electronic signature method and management
- Audit Trail
Since releasing a Part 11 compliant CISAS/EX medical monitoring system in 2003, we have since released Part 11 compliant products such as
the KR series of graphic recorder and
the MD8000 series of wireless loggers.
In 2015, we released the electronic record management package software
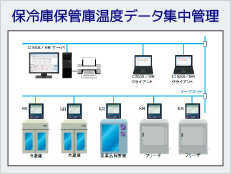
CISAS/ER, which allows for the centralized management of electronic record data measured by Part 11-compliant products of the KR series and MD8000 series, and we are now offering a system with excellent scalability and cost performance. All of our computer systems, recorder, and wireless loggers are Part 11 compliant, allowing us to meet a wide range of customer requirements.
ER/ES Guidelines
On April 1, 2005, the Ministry of Health, Labour and Welfare issued a notice from the Director-General of the Pharmaceuticals and Food Safety Bureau entitled "Use of Electromagnetic Records and Electronic Signatures in Applications for Approval or Licensing of Pharmaceuticals, etc." It is generally called the Ministry of Health, Labour and Welfare ER/ES Guidelines or ER/ES Guidelines. Electromagnetic records are a Japanese legal term and refer to electronic records. Although there are differences from Part 11 in terms of scope of application and electronic signatures, the basic content conforms to Part 11, and it is sometimes called the Japanese version of Part 11.
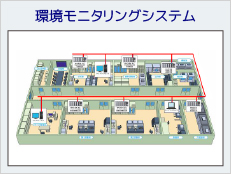
Part 11 compliant system example
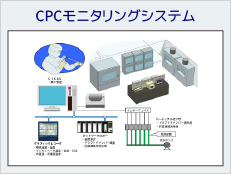
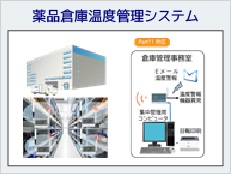
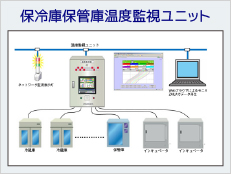
Part 11 compliant products
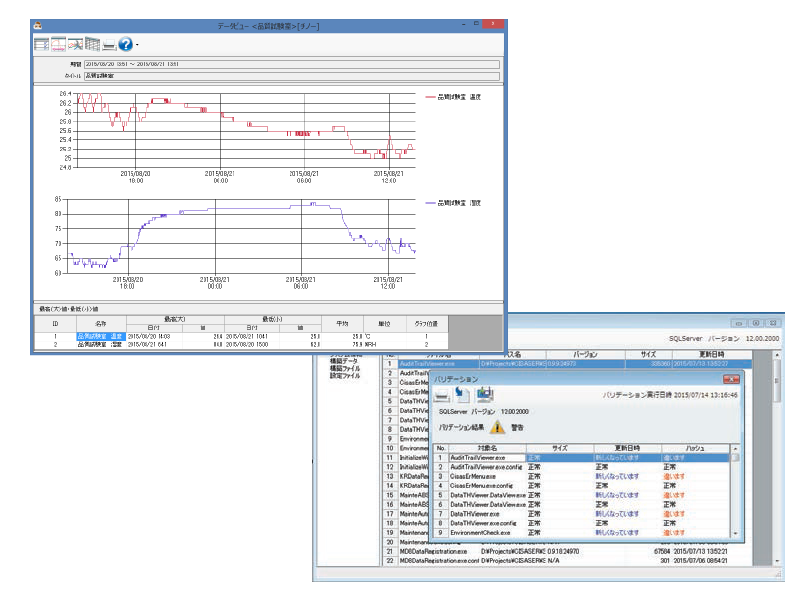
Electronic records management package software CISAS/ER
Centralized management of measurement data collected by Part 11-compliant wireless loggers and graphics recorder as electronic records.
Graphs by viewing data display, daily/monthly/report creation (printing, PDF output), and electronic signatures are possible.