Computerized System Validation (CSV)
Computerized System Validation (CSV)
Validation means confirmation of qualification, and involves "verifying that the equipment, procedure, and process required for pharmaceutical manufacturing and quality control produce the expected results" and "documenting" this.
Computer-based equipment and systems involve software such as operating systems and application, and their qualification is defined as computerized system validation.
Computerized system validation is also known as CSV, which is an abbreviation of “Computerized System Validation.”
To perform computerized system validation
One method for validating computerized systems is GAMP (Good Automated Manufacturing Practice), published by ISPE (International Society for Pharmaceutical Engineering), which serves as a global standard practice guideline.
The current GAMP is version 5.0, GAMP5 was published in 2008.
The guidance issued before that is referred to as GAMP4 to distinguish it.
In Japan, the Ministry of Health, Labor and Welfare issued a notice from the Director of the Surveillance and Guidance/Drug Control Division, Pharmaceuticals and Food Safety Bureau, entitled "Guidelines for the Proper Management of Computerized Systems for Drug and Quasi-Drug Manufacturers" (hereinafter referred to as "Proper Management Guidelines") on October 21, 2010, which is based on GAMP5 and came into effect on April 1, 2012. Depending on the regulatory requirements of your company, you may need to validate your computerized system in accordance with these guidances and guidelines.
We carry out validation of computerized systems in Japan in accordance with the guidelines for proper management, but we also carry out validation that meets the regulatory requirements of our customers.
Computerized system validation implementation and support
We use our extensive product lineup to propose computerized systems that are suited to customer requirements, and our experienced and proven specialist engineers carry out validation work, create deliverables, and support the creation of validation documents.
This is one of the items to be implemented in computerized system validation.
calibration We have been operating in this field for many years and have built up trust and a proven track record.
Regarding temperature and humidity, which are also important factors in the life sciences field,
JCSS Calibration We are a certified business operator and boast a high level of reliability.
Software categorization and implementation details
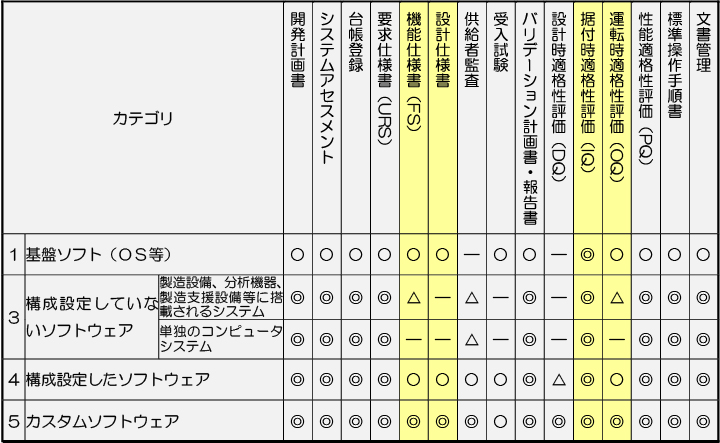
One of the key points in implementing computerized system validation is the categorization of the software, and the content of the validation varies depending on the category. The table below shows the category classifications given in the Guidelines for Appropriate Management and examples of general validation responses.
Category 3 "configuration software" is software whose configuration (functions, etc.) does not change depending on the settings for operation. Examples of our products that fall into this category include
the KR series of graphic recorder,
application software with security functions for wireless loggers,
the CISAS/V4 data acquisition and monitoring package system, and
the CISAS/ER electronic record management package software.
Category 4 "configuration software" refers to software configuration (functions, etc.) changes depending on the settings, and which can achieve complex functions by combining software modules.
In the Guidelines for Appropriate Management, as shown in the table, validation items to be implemented range from "development Plan" to "Document Management," and generally, the items to be implemented by suppliers are functional specifications (FS), design specifications, installation qualification (IQ), and operational qualification (OQ).
We will not only validate the items implemented by the supplier, but also provide technical support and document creation support for other items after consultation.
Category classification and general correspondence examples
◎: Required ○: Depends on the results of system assessment (basically required) △: Depends on the results of system assessment -: Optional