ISO15189 (Requirements for quality and competence of clinical laboratories)
ISO15189 is a international standards that sets out the requirements for the quality and competence of clinical laboratories. The first edition was established in 2003 and revised to the third edition in 2012. ISO 15189 (2012) is configuration in chapters 1~5 and two annexes and contains all the requirements for a clinical laboratory to certify that it operates a management system, is technically competent and capable of producing technically reasonable results.
Since ISO 15189 is international standards, test result reports can be accepted worldwide through international mutual recognition, and in the 2016 revision of medical fees, a new addition of 40 points was added as an "international standard laboratory management surcharge," which is a great advantage. In Japan, the "Japan Accreditation Association" is the only clinical laboratory accreditation body.
ISO 15189 accreditation for a laboratory can help standardize operations, elevate technicians, prove technical competence, and improve the reliability of results. In addition, with the enforcement of some of the provisions of the Act to Partially Amend the Medical Care Act, etc. (Act No. 57 of 29) on December 1, 2018, the Ministerial Ordinance on the Preparation of Ministerial Ordinances Related to the Ministry of Health, Labour and Welfare (Ministry of Health, Labour and Welfare Ordinance No. 93 of 2018) will also come into effect on December 1, 2018. The notice titled "Enforcement of the Ministerial Ordinance Concerning the Preparation of Ministerial Ordinances Related to the Ministry of Health, Labour and Welfare in Conjunction with the Enforcement of Part of the Act to Partially Amend the Medical Care Act, etc. (Medical Policy No. 0810 No. 1)" clearly states that it is desirable to strive to establish the system necessary for third-party accreditation of testing facilities such as ISO 15189 in order to ensure the accuracy of gene-related tests and chromosome tests.
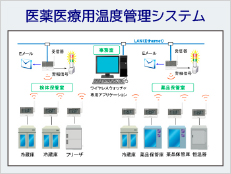
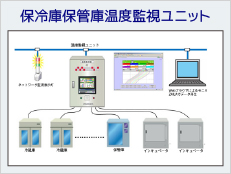
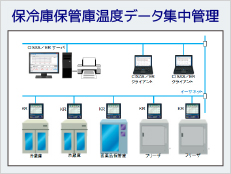
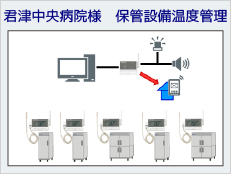
ISO15189 (2012) requires appropriate recording and management by controlling the temperature of refrigerators and freezers for storing specimens. In addition, temperature-control devices (e.g., incubators, refrigerators, freezers, etc.) must be monitored to ensure stability and uniformity of temperature. For temperature control, measuring instruments are also required to be quality controlled, and regular calibration and inspection are required.
System Example
Applicable products and services
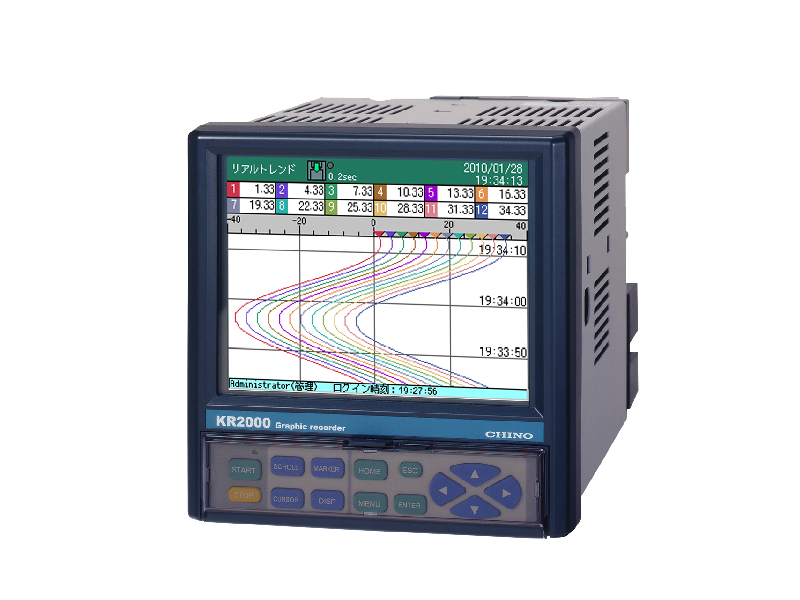
Graphic recorder with measurement data protection function KR2P00
Paperless recorder with 6 and 12 input points
Although it is small, it has a wide range display and high performance, making it suitable for use as a standalone device or small-scale monitoring equipment.
When combined with the CISAS series, it can be configured as a recorder for a computer system.
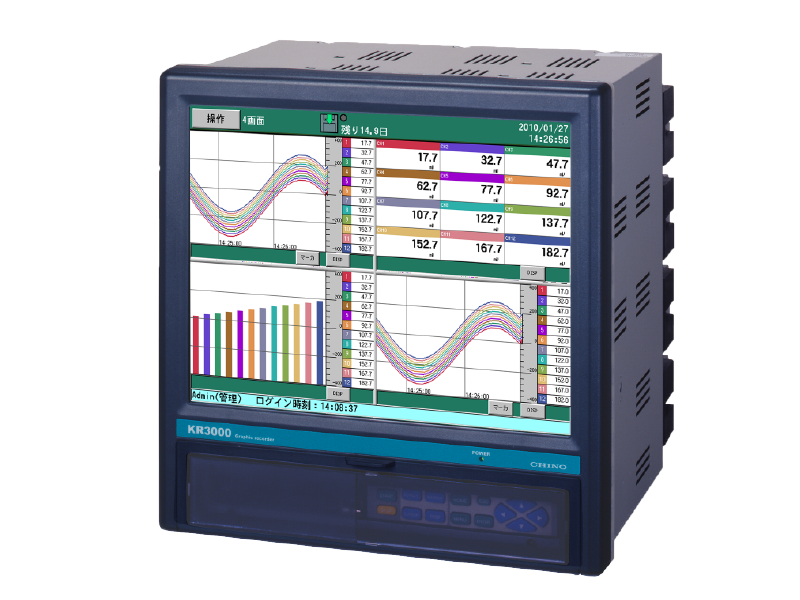
Graphic recorder with measurement data protection function KR3P00
Paperless recorder with up to 48 input points
With its versatile display and high performance, it is suitable for data management on equipment such as stability testers, refrigerators, and freezers.
When combined with the CISAS series, it can be configured as a recorder for a computer system.
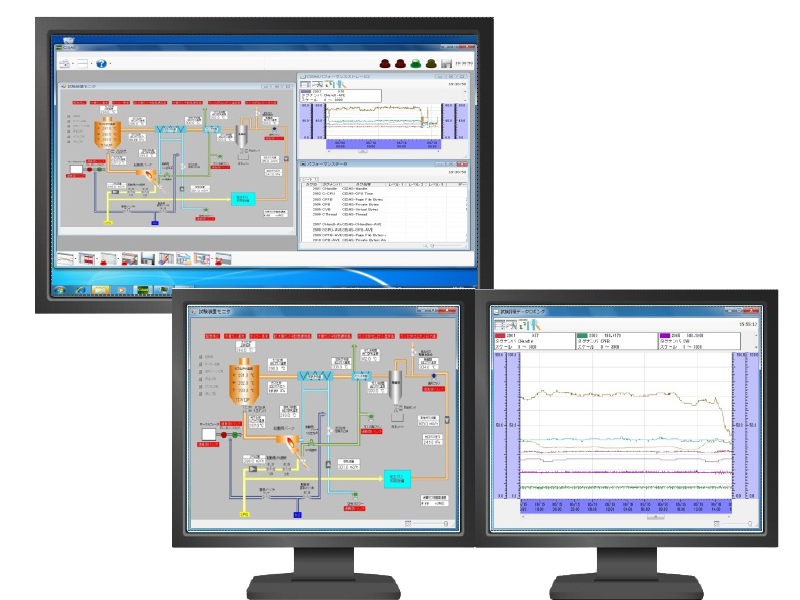
Recording and monitoring package system CISAS/V4
CISAS/V4 is a package system that uses our recorders, loggers, and controllers, as well as commercially available PLCs (programmable controllers), as system components to collect and monitor data from up to 5,000 tags of various devices and equipment on a PC.
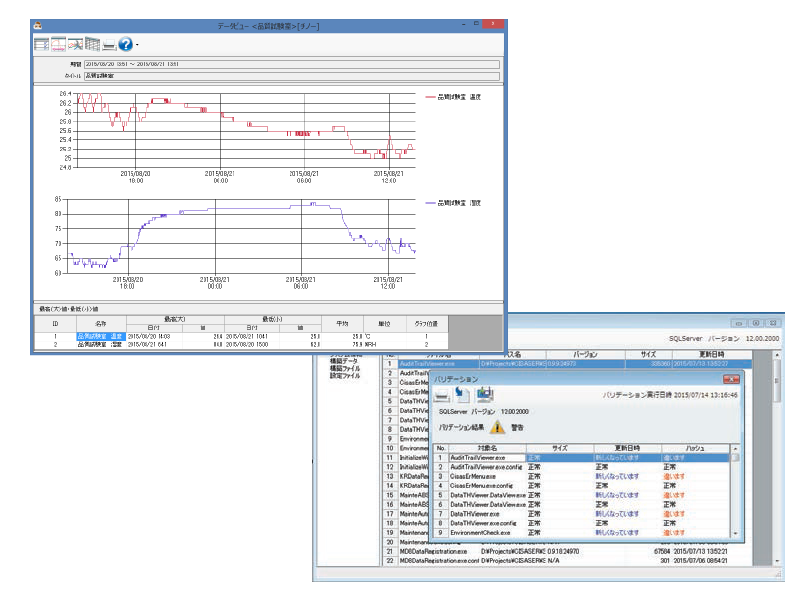
Electronic records management package software CISAS/ER
Measurement data recorded by Part 11-compliant wireless loggers and graphic recorder can be centrally managed as electronic records.
Graph display by viewing data, daily reports, monthly reports, and reports can be created (printed and output as PDF files) and electronic signatures are possible

JCSS (logo mark) calibration test
We are registration as a calibration laboratory for temperature and humidity under the Measurement Law.
We have also acquired certification as an "MRA-compliant certified business operator" based on ISO/IEC17025, and can issue calibration certificates with the JCSS certification symbol mark.

On-site inspection and calibration services
Our service technicians or contracted service personnel visit the user's site to inspect and calibrate measuring instruments, mainly temperature and humidity measuring instruments, regulators, recorders, etc.